1.1 – Measurements in physics
- Fundamental and derived units
Fundamental SI units
| Quantity | SI unit | Symbol |
| Mass | Kilogram | kg |
| Distance | Meter | m |
| Time | Second | s |
| Electric current | Ampere | A |
| Amount of substance | Mole | mol |
| Temperature | Kelvin | K |
Derived units are combinations of fundamental units. Some examples are:
- m/s (Unit for velocity)
- N (kg*m/s^2) (Unit for force)
- J (kg*m^2/s^2) (Unit for energy)
- Scientific notation and metric multipliers
In scientific notation, values are written in the form a*10^n, where a is a number within 1 and 10 and n is any integer. Some examples are:
- The speed of light is 300000000 (m/s). In scientific notation, this is expressed as 3*10^8
- A centimeter (cm) is 1/100 of a meter (m). In scientific notation, one cm is expressed as 1*10^-2 m.
Metric multipliers
| Prefix | Abbreviation | Value |
| peta | P | 10^15 |
| tera | T | 10^12 |
| giga | G | 10^9 |
| mega | M | 10^6 |
| kilo | k | 10^3 |
| hecto | h | 10^2 |
| deca | da | 10^1 |
| deci | d | 10^-1 |
| centi | c | 10^-2 |
| milli | m | 10^-3 |
| micro | μ | 10^-6 |
| nano | n | 10^-9 |
| pico | p | 10^-12 |
| femto | f | 10^-15 |
- Significant figures
For a certain value, all figures are significant, except:
- Leading zeros
- Trailing zeros if this value does not have a decimal point, for example:
- 12300 has 3 significant figures. The two trailing zeros are not significant.
- 012300 has 5 significant figures. The two leading zeros are not significant. The two trailing zeros are significant.
When multiplying or dividing numbers, the number of significant figures of the result value should not exceed the least precise value of the calculation.
The number of significant figures in any answer should be consistent with the number of significant figures of the given data in the question.
FYI
- In multiplication/division, give the answer to the lowest significant figure (S.F.).
- In addition/subtraction, give the answer to the lowest decimal place (D.P.).
- Orders of magnitude
Orders of magnitude are given in powers of 10, likewise those given in the scientific notation section previously.
Orders of magnitude are used to compare the size of physical data.
| Distance | Magnitude (m) | Order of magnitude |
| Diameter of the observable universe | 10^26 | 26 |
| Diameter of the Milky Way galaxy | 10^21 | 21 |
| Diameter of the Solar System | 10^13 | 13 |
| Distance to the Sun | 10^11 | 11 |
| Radius of the Earth | 10^7 | 7 |
| Diameter of a hydrogen atom | 10^-10 | 10 |
| Diameter of a nucleus | 10^-15 | 15 |
| Diameter of a proton | 10^-15 | 15 |
| Mass | Magnitude (kg) | Order of magnitude |
| The universe | 10^53 | 53 |
| The Milky Way galaxy | 10^41 | 41 |
| The Sun | 10^30 | 30 |
| The Earth | 10^24 | 24 |
| A hydrogen atom | 10^-27 | -27 |
| An electron | 10^-30 | -30 |
| Time | Magnitude (s) | Order of magnitude |
| Age of the universe | 10^17 | 17 |
| One year | 10^7 | 7 |
| One day | 10^5 | 5 |
| An hour | 10^3 | 3 |
| Period of heartheart | 10^0 | 0 |
- Estimation
Estimations are usually made to the nearest power of 10. Some examples are given in the tables in the orders of magnitude section.
1.2 – Uncertainties and errors
- Random and systematic errors
| Random error | Systematic error |
| Caused by fluctuations in measurements centered around the true value (spread). Can be reduced by averaging over repeated measurements. Not caused by bias. | Caused by fixed shifts in measurements away from the true value. Cannot be reduced by averaging over repeated measurements. Caused by bias. |
| Examples: Fluctuations in room temperature The noise in circuits Human error | Examples: Equipment calibration error such as the zero offset error Incorrect method of measurement |

- Absolute, fractional and percentage uncertainties
Physical measurements are sometimes expressed in the form x±Δx. For example, 10±1 would mean a range from 9 to 11 for the measurement.
| Absolute uncertainty | Δx |
| Fractional uncertainty | Δx /x |
| Percentage uncertainty | Δx/x*100% |
Calculating with uncertainties
| Addition/Subtraction | y=a±b | Δy=Δa+Δb (sum of absolute uncertainties) |
| Multiplication/Division | y=a*b or y=a/b | Δy/y=Δa/a+Δb/b (sum of fractional uncertainties) |
| Power | y=a^n | Δy/y=|n|Δa/a (|n| times fractional uncertainty) |
- Error bars
Error bars are bars on graphs which indicate uncertainties. They can be horizontal or vertical with the total length of two absolute uncertainties.

- Uncertainty of gradient and intercepts
Line of best fit: The straight line drawn on a graph so that the average distance between the data points and the line is minimized.
Maximum/Minimum line: The two lines with maximum possible slope and minimum possible slope given that they both pass through all the error bars.

The uncertainty in the intercepts of a straight line graph: The difference between the intercepts of the line of best fit and the maximum/minimum line.
The uncertainty in the gradient: The difference between the gradients of the line of best fit and the maximum/minimum line.
1.3 – Vectors and scalars
- Vector and scalar quantities
| Scalar | Vector |
| A quantity which is defined by its magnitude only. | A quantity which is defined by both is magnitude and direction. |
| Examples: Distance Speed Time Energy | Examples: Displacement Velocity Acceleration Force |
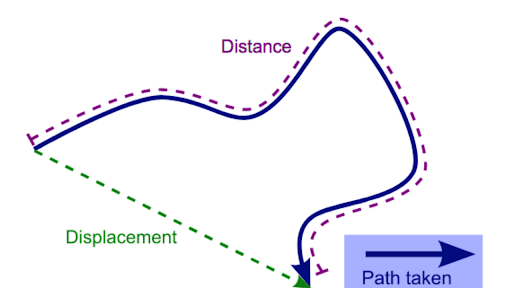
- Combination and resolution of vectors
Vector addition and subtraction can be done by the parallelogram method or the head to tail method. Vectors that form a closed polygon (cycle) add up to zero.

When resolving vectors in two directions, vectors can be resolved into a pair of perpendicular components.

FYI
The relationship between two sets of data can be determined graphically.
| Relationship | Type of Graph | Slope | y-intercept |
| y=mx+c | y against x | m | c |
| y=kx^n | logy against logx | n | logk |
| y=kx^n+c with n given | y against x^n | k | c |
